 Image credit: Cancer Research Communications
Image credit: Cancer Research Communications
Abstract
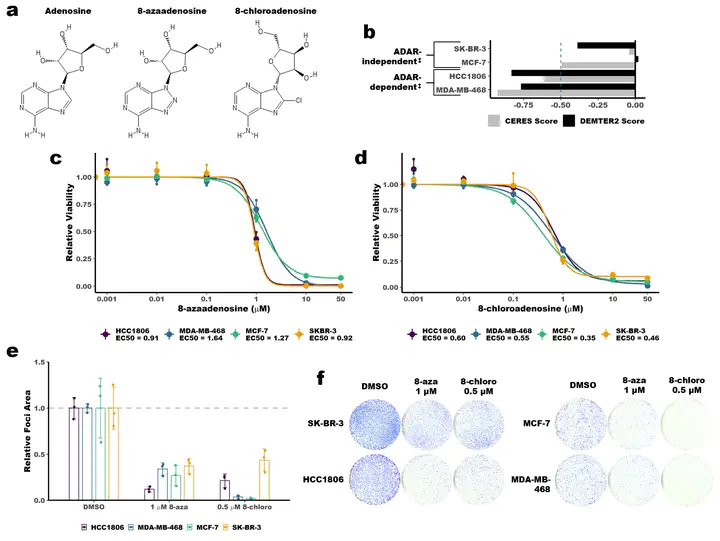
The RNA editing enzyme ADAR is an attractive therapeutic target for multiple cancers. Through its deaminase activity, ADAR edits adenosine to inosine in double-stranded RNAs. Loss of ADAR in some cancer cell lines causes activation of the type I IFN pathway and the PKR translational repressor, leading to inhibition of proliferation and stimulation of cell death. As such, inhibition of ADAR function is a viable therapeutic strategy for many cancers. However, there are no FDA-approved inhibitors of ADAR. Two small molecules have been previously shown to inhibit ADAR or reduce its expression - 8-azaadenosine and 8-chloroadenosine. Here we show that neither molecule is a selective inhibitor of ADAR. Both 8-azaadenosine and 8-chloroadenosine show similar toxicity to ADAR-dependent and -independent cancer cell lines. Furthermore, the toxicity of both small molecules is comparable between cell lines with either knockdown or overexpression of ADAR, and cells with unperturbed ADAR expression. Treatment with neither molecule causes activation of PKR. Finally, treatment with either molecule has no effect on A-to-I editing of multiple ADAR substrates. Together, these data show that 8-azaadenosine and 8-chloroadenosine are not suitable small molecules for therapies that require selective inhibition of ADAR, and neither should be used in preclinical studies as ADAR inhibitors.